ENTEC in Horticulture - Nitrogen stabilisers in the soil
Aug 11, 2020Horticulture crops need nitrogen (N) to consistently produce high yielding and exceptional quality crops.
The challenge is to select a nitrogen fertiliser product which will reduce risk, improve nutrient use efficiency, optimise production, and benefit quality.
Not all fertilisers perform equally. This is where ENTEC®, an ammonium stabiliser and enhanced efficiency fertiliser, has a great fit.
Fertiliser product choice starts with understanding the N cycle and potential losses (Figure 1). Nitrogen in fertiliser, crop residues, manure or compost is transformed by soil microorganisms either into an organic form (soil organic matter) or into a plant-available form (nitrate).
Over time, any nitrogen immobilised into organic matter can become plant-available later. This is only a temporary loss. Two other losses – denitrification and leaching – are more costly.
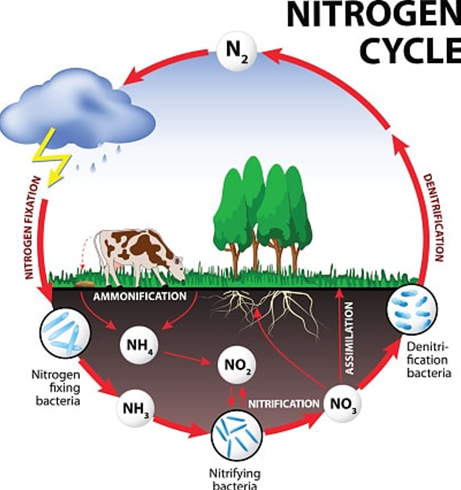
Figure 1: The Nitrogen cycle.
Nitrogen Sources
Soil N supply from organic matter
Mineralisation of soil organic matter is a significant contributor to crop N supply. Mineralisation involves soil microorganisms transforming the ‘non-plant available’ organically-bound N into ‘plant available’ mineral N. Any nitrogen associated with the likes of crop residues, fallow crops or composts, also enters the pool of organically-bound N.
Soil N supply from fertilisers
Soil testing is important in horticulture. Testing tells the difference between what ‘soil fertility’ can provide crops naturally, compared to the amount of nutrients crops require to flourish. The type and amount of nutrient differences between these ‘crop required’ & ‘soil fertility’ levels can be applied as fertilisers.
Nitrogen Losses
Immobilisation
Just as N is subject to biological transformation to be made ‘plant-available’ under certain soil conditions, a similar biological transformation can also tie-up nutrients. This is known as immobilisation, and it is considered to be a short to medium term N loss pathway. Over time, this N may become plant available again.
Nitrogen conversion chemistry - nitrification and leaching
During the process of nitrification, nitrogen is converted by Nitrosomonas soil bacteria from Ammonium (NH4+) into Nitrite (NO2-) and then into Nitrate (NO3-) by another group of soil bacteria, Nitrobacter:
NITRIFICATION
Ammonium (NH4+) ------------------ Nitrite (NO2-) --------------- Nitrate (NO3-)
Nitrosomonas Nitrobacter
In terms of loss pathways, the issue with NO3- is that it can be lost via both leaching and denitrification. Leaching takes nitrate through the soil & out of the root zone. Denitrification turns nitrate into gasses which are then lost from the soil to the atmosphere.
The reason NO3- leaching occurs is due to its negative charge, i.e. soil colloids and soil organic matter possess a negative charge, hence two negatives cannot attract. So as water moves through lighter soils, NO3- moves with it. There is a fine balance at play here – between water & NO3-. We need to regulary irrigate, to keep the crops productive. Then rainfall events occur also. However, every time water moves through the soil, NO3- is also moving with it. If nitrate moves beyond the root zone, crops can not access it to grow. Leaching is common in soils with a cation exchange capacity (CEC) of <4 cmol(+)/kg.
Denitrification occurs due to the microbes in soil, as they start running out of oxygen in wet to saturated conditions (i.e. 60% or greater water filled pore space). This can be worsened by poorly structured or compacted soils, where high soil organic matter levels create high soil microbe populations, with over irrigation or when experiencing significant rainfall events. Under these conditions, the microbes use NO3- as an oxygen supply. The conversion is as follows:
NO3- to NO2- to NO to N2O to N2
(Nitrate) (Nitrite) (Nitric Oxide) (Nitrous oxide*) (Di-nitrogen)
* A potent green house gas, 298 time worse than carbon dioxide (CO2).
Why ENTEC®?
Entec is an ammonium stabiliser – it slows the conversion rate of ammonium to nitrate. It works by inhibiting the Nitrosomonas group of soil bacteria which keeps N in the top soil in the ammonium form (NH4+) for longer. As ammonium it has a positive charge, and therefore, will be not readily leach through the profile and has a zero denitrification loss potential. Crops can still take up N when it is in the ammonium form.
ENTEC also reduces nitrous oxide emissions. In Victorian trials, ENTEC Nitrophoska was shown to reduce the average nitrous oxide N2O emission flux by 69 to 100% compared to traditional fertilisers (Melville et al. 2012).
This means ENTEC keeps more nitrogen where you want it most – in the soil and available to the crop.
In a horticulture situation, seedlings or transplants do not use a lot of nitrogen during early establishment. Using ENTEC with the base fertiliser helps to reduce these nitrogen losses by keeping the N in the more stable and still plant-available ammonium form. As the crop becomes established, there is N available in the root zone for crop uptake.
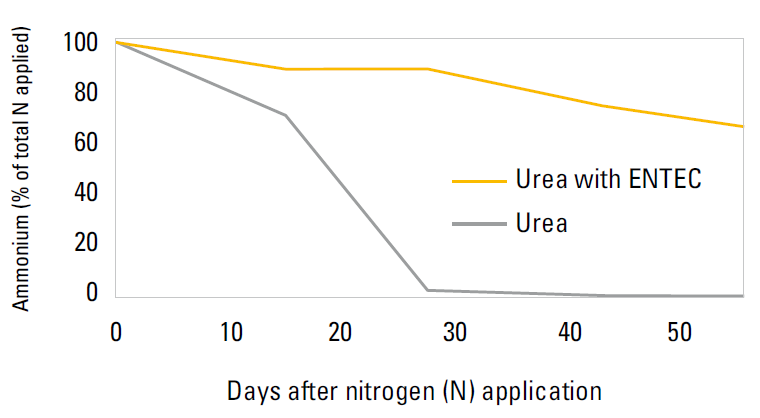
ENTEC has been shown to be effective in the soil from 4 – 10 weeks. This has been demonstrated in laboratory results in a moist alkaline vertosol soil at 250C, 97% of applied fertiliser had converted to nitrate after 14 days without ENTEC protection. When ENTEC was used, over 70% of the nitrogen was stabilised as ammonium after 50 days (Suter et al. 2008).
This is very important for crops grown under irrigation. For example, for an irrigated broccoli crop where there is crop residue, wet conditions (due to overhead irrigation), compacted soil and warm temperatures, the ENTEC blend improved yield (Table 1).
Using NH4+ stabilisers ensures flexibility regarding timing of application. Fertiliser application can be done early in crop development, without compromising yield N loss potentials. Potentially fewer fertiliser applications can be required when using an NH4+ stabiliser, with more N being protected and ‘banked’ in the topsoil. Melville et al. (2012) demonstrated that one fertiliser application could be dropped in Victoria due to enhanced N use efficiency.
|
Crop |
Location |
Product/s |
ENTEC |
Conventional |
Percentage Yield |
Statistics |
|
|||||
|
|
||||||||||||
|
|
||||||||||||
|
Potato |
Atherton |
Entec CK-800 v CK-800 |
39.25 t/ha |
34.95 t/ha |
12.3% |
LSD 3.79 |
|
|||||
|
Potato |
Atherton |
Entec Nitrophoska v Nitrophoska |
36.55 t/ha |
32.10 t/ha |
13.9% |
LSD 3.79 |
|
|||||
|
Potato |
Pinnaroo, 2016 |
Entec Nitrophoska v Nitrophoska |
73.05 t/ha |
59.65 t/ha |
22.5% |
LSD 10.55 |
|
|||||
|
Broccoli |
Boneo, 2012 |
Entec Nitrophoska v Nitrophoska |
4.98 kg/plot |
3.23 kg/plot |
58.6% |
LSD 1.29 |
|
|||||
|
|
||||||||||||
|
|
||||||||||||
|
Broccoli |
Boneo, 2012 |
Entec Nitrophoska v Nitrophoska |
5.55 kg/plot |
3.14 kg/plot |
76.8% |
LSD 1.29 |
|
|||||
|
|
||||||||||||
|
|
||||||||||||
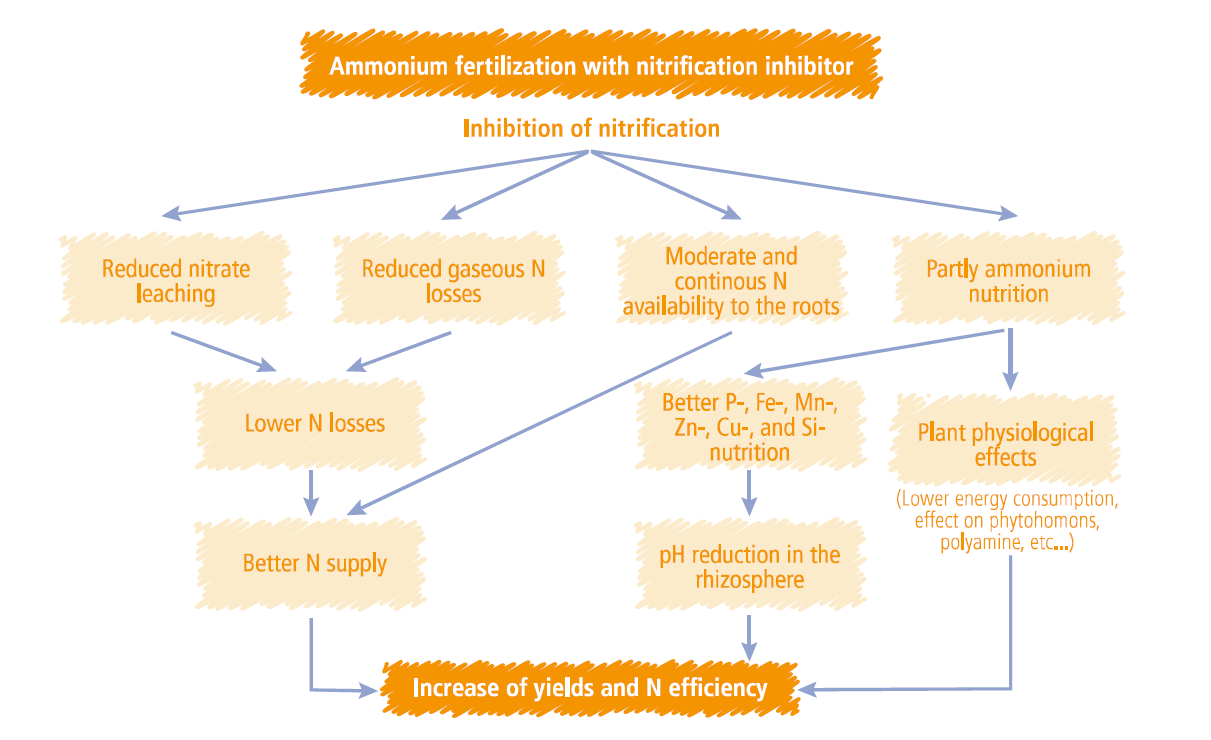
There is more to ENTEC than just reducing leaching & denitrification:
Crop physiological benefits
Advantages of Ammonium nutrition
References:
- Hähndel R, Lang H, Hermann P (1994) Wirkung von Ammoniumstabilisierten Düngern auf Ertrag und Qualität von Gemüse. Agribiol Res 47:101–108.
- Buerkert B, Horlacher D, Marschner H (1995) Time course of nitrogen in soil solution and nitrogen uptake in maize plants as affected by form of application and application time of fertilizer nitrogen. J Agron Crop Sci 174:325–336.
- Burghardt H, Ellering K (1988) Beeinflussung des Nitratgehaltes von Spinat durch unterschiedliche Kulturbedingungen. Gartenbauwissenschaft 53:201–205.
- Müller H, Wedler A (1987) Ein Beitrag zum Einsatz des Nitrifkationshemmstoff Dicyandiamid (DCD) zur Verminderung des Nitratgehaltes in Gemüse. Landwirtsch Forsch 40:78–87.
- Venter F (1984) Nitrate contents of head lettuce as influenced by nitrificides. Acta Hortic 163:231–236.
- Slangen JHG, Kerkhoff P (1984) Nitrification inhibitors in agriculture and horticulture: a literature review. Fert Res 5:1–76.
- Ali IA, Kafkafi U, Ymaguchi I, Sugimoto Y, Inanaga S (1998) Response of oilseed rape plant to low root temperature and nitrate: ammonium ratios. J Plant Nutr 21:1463–1481.
-
Gerendás J, Sattelmacher B (1995) Einfluß des Ammoniumangebotes auf Wachstum, Mineralstoff- und Polyamingehalt.
Junger Maispflanzen. Z Pflanzenernaehr Bodenkd 158:299–305. - Klein, H, Priebe, A., & Jager, H. (1979) Putrescine and Spermidine in Peas: Effects on Nitrogen Source and Potassium Supply. Physiologia Plantarum, 45: 497-499.
- Melville et al. (2012) Carbon and Sustainability. A demonstration on vegetable properties across Australia.
- Pasda, G., Handel, R. and Zerulla, W (2001) Effects of fertilizers with the new nitrogen inhibitor DMPP (3,4-dimethlypyrazole phosphate) on yield and quality of agricultural and horticultural crops. Biological Fertility Soils, 34, 85-97
- Jing et al. (2010) Localized application of phosphorus and ammonium improves growth of maize seedlings by stimulating root proliferation and rhizosphere acidification. Field Crops Research. 119. 355-364.
- Ulrich, W. (1992) Transport of nitrate and ammonium through plant membranes. In: Mengel K. Pilbeam D. (eds) Nitrogen metabolism of plants. Oxford Science, Clarendon press, Oxford, pp 121-137.
- Smiciklas KD, Below FE (1992) Role of cytokinin in enhanced productivity of maize supplied with NH4+ and NO3-. Plant Soil. 142:307–313.
- ‘Ammonium uptake by rice roots’ Wang, M., Glass, A., Shaff, J. and Kochian, L. (1994) III. Ectrophysiology. Plant Physiology, 104, 899-906.
- Wang X, Below FE (1996) Cytokinins enhanced growth and tillering of wheat induced by mixed nitrogen source. Crop Sci. 36:121–126.
- Trolldenier G (1981) Influence of soil moisture, soil acidity and nitrogen source on take-all of wheat. Phytopathol Z 102: 163–177.
